Assessing the Total Economic Burden of Rare Disease
Summary
Innovative research methods can be used to measure the total burden of rare disease, including direct medical costs, indirect costs, and overall life impacts.In this whitepaper, Avalere describes the opportunity and importance of measuring the total economic impact of diseases, particularly for rare diseases. First, experts present a case for considering the total economic burden of rare disease, then explore the current state of evidence on the burden of rare diseases, concluding with real-world application of total economic impact research. The proposed hybrid study approach builds upon a prior assessment by the EveryLife Foundation on the economic burden of 379 rare diseases, which can be used as a springboard for studying the disease-specific social and economic burden of a single rare disease. The team selected generalized myasthenia gravis (gMG) as a case study to measure direct and indirect non-medical costs based on Avalere’s foundational research on patient- and caregiver-centered life impacts. A previous mixed-methods analysis found that financial and occupational impacts ranked highly among the eight impacts studied—emotional, financial, occupational, physical, planning & autonomy, safety, sleep, and social—elevating the importance of economic impacts.
Figure 1 details potential components of total economic burden to be studied to better understand the true cost of these conditions, such as the significant out-of-pocket costs, labor and productivity impacts, and broader societal impacts. Understanding the true burden of rare diseases can help policymakers and the public better recognize the extent of the individual, family, and social impacts and allocate resources accordingly.
Figure 1: Potential Components of a Total Economic Burden Study for Rare Diseases
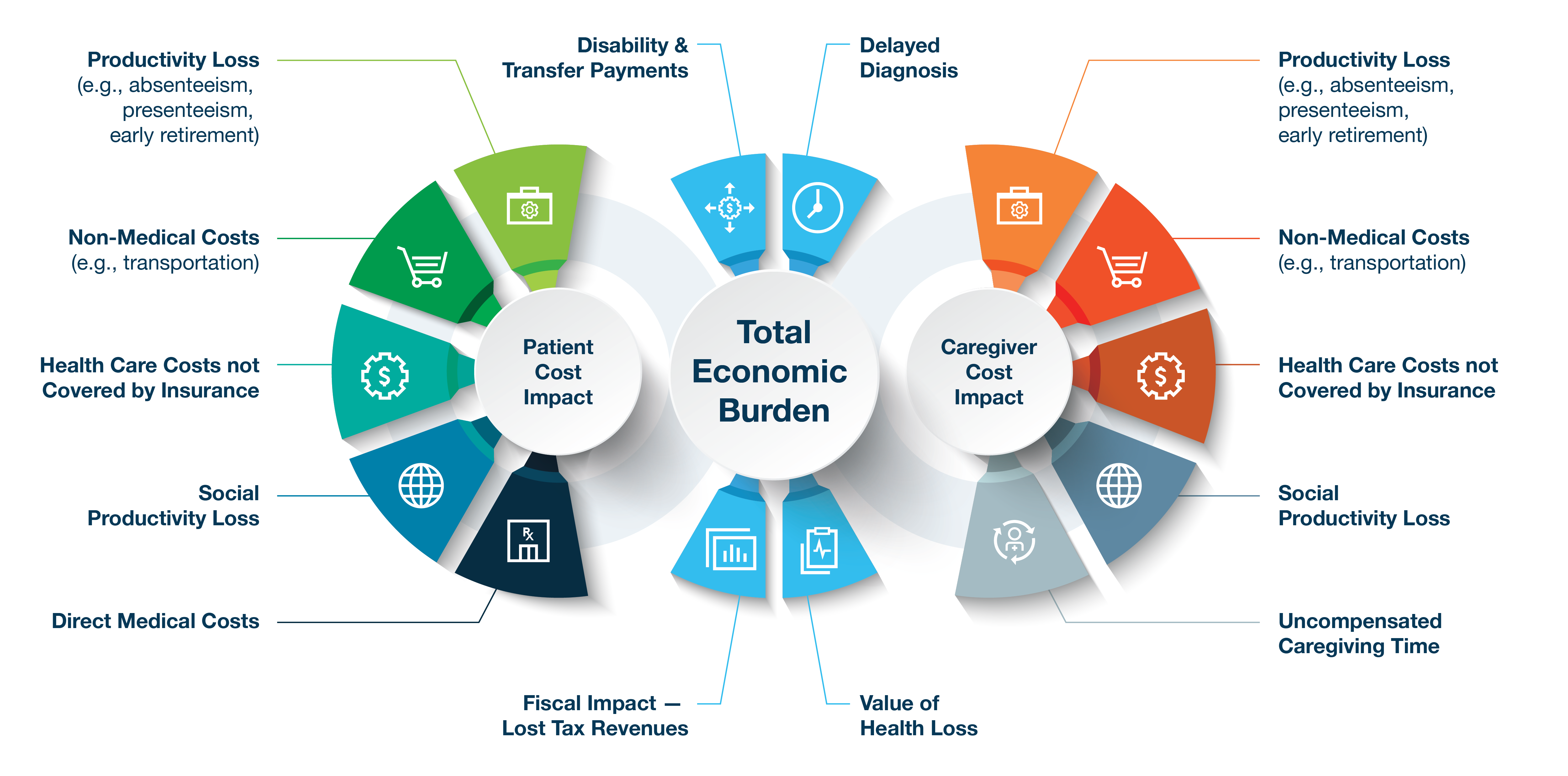
Despite the challenges of calculating the total burden of disease (especially for heterogeneous populations), robust and well-designed research can offer useful insights and context for patients, caregivers, payers, health technology assessment bodies, healthcare providers, policymakers, manufacturers, researchers, and society.
Access the whitepaper here.
Funding for this whitepaper was provided by Alexion, AstraZeneca Rare Disease. Avalere Health retained full editorial control.





