Avalere Analysis Finds Exchange Coverage of Drugs Used to Treat Patients with Rare Diseases Varies
Summary
A new analysis from Avalere Health, published today in the "Journal of Managed Care & Specialty Pharmacy," examined exchange coverage rates of 11 drugs used to treat rare diseases and found that plans in the exchange cover these drugs 65 percent of the time on average across plans, although coverage varies widely by product and metal level.The analysis included bronze- and silver-level exchange plans in the 15 largest states. The selected drugs are used to treat Huntington disease, hydatidosis, parathyroid carcinoma, atypical hemolytic uremic syndrome, sickle cell anemia, advanced soft tissue sarcoma, and Gaucher disease type 1.
Coverage of the drugs analyzed varied considerably with bronze plans less likely to cover the selected drugs than silver plans. Plan use of utilization management, such as prior authorization (PA), step therapy (ST) or a combination of both, varied widely by drug from 6 percent for Albenza (hydatidosis) to nearly 75 percent of plans for Xenazine (Huntington disease). The rare disease drugs were frequently placed on the highest tier of 4-tier formularies with more than 70 percent of plans using coinsurance for the highest tiers. Coinsurance ranges from 10-50% in silver plans.
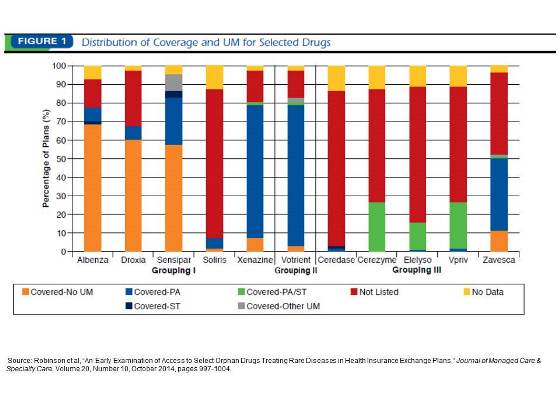
Finally, the analysis showed that the process patients go through to select a plan may be complicated by how plans cover certain medications as part of either the medical or pharmacy benefit. Products that are typically physician administered (such as Soliris, Ceredase, Cerezyme, Elelyso, and Vpriv) are most often covered under a plan’s medical benefit and are “not listed” (as indicated above) on plan drug formularies. “This analysis highlights the need for increased data transparency in the exchanges so that patients with rare diseases can make more informed plan selections,” said Sandy Robinson, the study’s lead author and senior vice president at Avalere Health.
The out of pocket maximum of $6,350 for an individual offers some financial protection for patients with high health expenses. These patients also meet their maximum out of pocket cost earlier in the year, often incurring the full amount in the first or second month of therapy.
The analysis was published by the Journal of Managed Care & Specialty Pharmacy on September 22, 2014. Full text of the article is available here.
This analysis was funded by Novartis Pharmaceuticals. The authors maintained editorial control.
Methodology
Avalere Planscape™ evaluated 84 formularies from bronze- and silver-level plans in 15 largest states. Avalere analyzed 2014, exchange-specific formularies publicly available at the time of this analysis (November 2013). Avalere captured all coverage, tiering, and utilization management data available in public documents.
For more information about rare disease drug coverage in the exchanges, contact Lauren Barnes at LBarnes@Avalere.com.



