Incorporating Clinical Factors Could Improve OCM Cost Prediction
Summary
An Avalere analysis found that among high-risk breast cancer episodes, those in later stages of the disease had higher episode expenditures relative to their benchmarks compared to those in earlier stages.The Oncology Care Model (OCM) is an episode-based, oncology-focused payment model from the Centers for Medicare & Medicaid Services (CMS) that aims to improve care coordination and reduce costs for Medicare fee-for-service (FFS) beneficiaries. The model evaluates the total cost of care for 6-month episodes initiated by chemotherapy treatments that are attributed to oncology practices voluntarily participating in the model. In each performance period (PP), the CMS measures practice performance based on their total expenditures relative to a benchmark price. The practice’s elected risk track for a PP, relative to their performance, determines if a practice earns a performance-based payment (PBP) or owes recoupment.
The basis for the benchmark price calculation is the OCM prediction model, which estimates episode costs based on key factors such as tumor type, beneficiary demographics and comorbidities, and utilization of certain services. While OCM participants are required to submit clinical data through a registry, these data were not originally used to set benchmark prices. However, beginning in PP7 (July 2, 2019), the CMS introduced a “metastatic adjuster” for breast, lung, and small intestine/colorectal cancers to factor metastatic disease into the benchmark price calculation. To better understand these dynamics, Avalere evaluated cost patterns within high-risk breast cancer episodes and the influence of the metastatic adjustor on episode expenditure and benchmark price. Avalere used claims-based indicators to group earlier-stage (stages I and II) and later-stage (stages III and IV) episodes. Avalere then replicated the OCM methodology and calculated each group’s episode expenditure and benchmark price in PP3 and PP4. Note: Avalere’s classification of earlier-stage and later-stage groups are estimates, as only the Center for Medicare & Medicaid Innovation has access to OCM clinical registry data.
Avalere found differential results between later- and earlier-stage episode subgroups. Results indicate that, on average, episode expenditures exceeded benchmark prices by about 10% for later-stage episodes. However, average episode expenditures were lower than benchmark prices by about 17% for earlier-stage episodes. While later-stage episodes had slightly higher episode expenditures relative to earlier-stage episodes, benchmark prices for earlier-stage episodes were notably higher than later-stage episodes (Figure 1). Therefore, through the first 6 performance periods, having a higher proportion of later-stage high-risk breast cancer episodes may have hindered practices’ ability to earn PBPs.
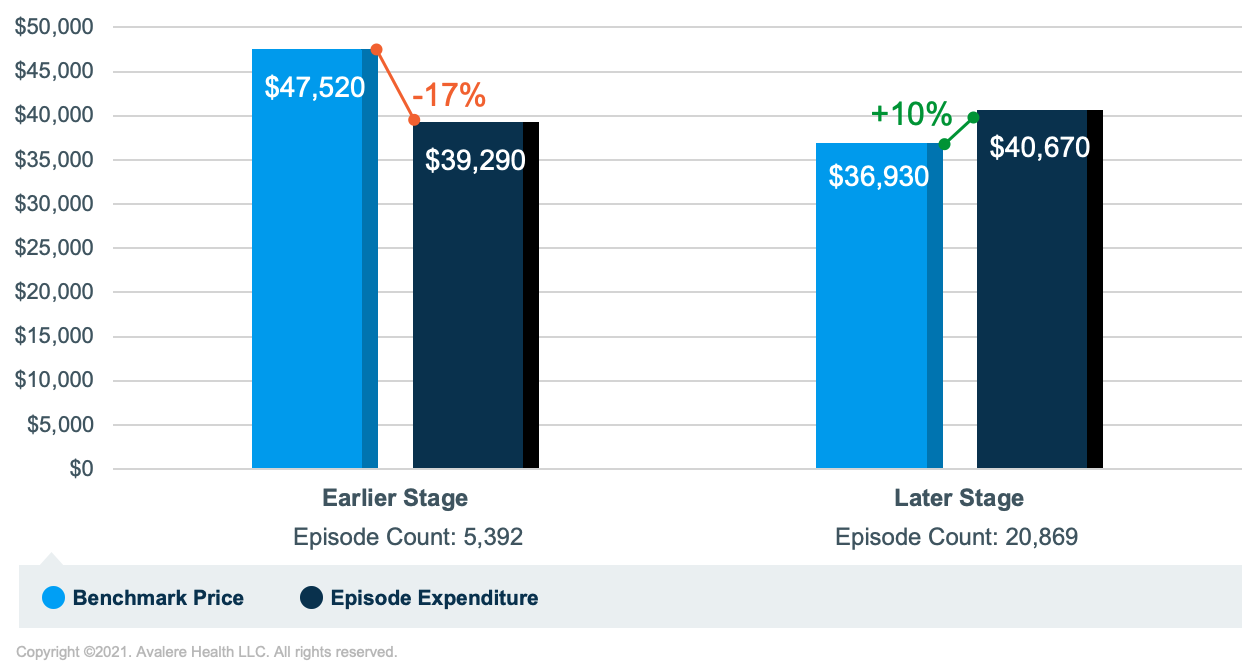
The disparity between benchmark prices for earlier-stage and later-stage breast cancer episodes partially results from the different treatments provided to beneficiaries and how the prediction model accounts for such treatments. Beneficiaries in earlier-stage episodes are more likely to receive surgery and radiation procedures than later-stage episodes. While episode expenditures for earlier- and later-stage episodes are similar, treatment costs in earlier-stage episodes (i.e., chemotherapy, radiation, and surgery) make up a smaller proportion of total episode expenditures compared to later-stage episode treatment costs. The prediction model adjusts for surgery and radiation treatment costs, as well as a history of prior chemotherapy use, but it does not account for other types of treatments. Accordingly, such adjustments may be increasing predictions for earlier-stage episodes and decreasing predictions for later-stage episodes.
Starting in PP7, the incorporation of a metastatic adjuster will mark the first time clinical registry data is factored into setting benchmark prices. Performance in metastatic episodes is likely to improve, and the adjuster could benefit practices with a higher proportion of later-stage breast, lung, and small intestine/colorectal episodes (the metastatic adjuster only applies to these 3 cancer types). As providers and payers continue to pursue episodic-based care in oncology and as the OCM evolves into its next iteration, the Oncology Care First model, it is important to understand how such models could leverage more clinical data to improve risk adjustment and cost predictions.
Funding for this work was provided in part by Genentech, a member of the Roche Group.
Methodology
Avalere performed this analysis using Medicare Part A/B FFS claims and Part D prescription drug event data under a CMS research data use agreement. The total sample of beneficiaries observed in this analysis does not exceed 20% of total Medicare beneficiaries. Avalere replicated the OCM payment methodology developed by the CMS, including the attribution of episodes to practices, the assignment of cancer type, and the calculation of benchmark prices. Participants were identified in the claims data through the tax identification numbers (TIN) reported on Part B claims. Avalere identified 175 practices that were ever enrolled in the OCM through TIN mapping and general research of oncology practices. The baseline period includes episodes initiated between January 1, 2012, and December 31, 2014, and the intervention period includes episodes initiated between July 2, 2017, and July 1, 2018 (PP3-PP4).
Avalere used the CMS’s definition of high-risk breast cancer, defining it as episodes that contain a chemotherapy treatment other than a long-term oral endocrine therapy. Avalere developed claims logic to determine which episodes were more likely earlier stage (stages I and II) and which were later stage (stages III and IV). Later stage episodes were defined as those that had either the presence of a metastasis ICD-10 diagnosis code or used certain drugs approved for stage III/IV disease and did not have the occurrence of radiation or surgery treatment within the episode.
To receive Avalere updates, connect with us.







